| Jean-Christophe Daigle and Jerome P. Claverie report a method for fabricating materials composed of inorganic nanoparticles encased in organic polymer shells. |
Reviewed by Dr. Michael Steigerwald, Columbia University
Daigle and Claverie report an interesting, versatile, and potentially valuable method for the large-scale fabrication of composite nanomaterials: inorganic nanoparticles encased in organic polymer shells. Starting with commercially available inorganic nanoparticulate materials, they first conduct a particle-attached polymerization to solubilize the particles and then a second polymer-attached polymerization to construct the organic shell. The most clever feature of the authors' method is the use of Reversible Addition Fragmentation chain Transfer (RAFT) as the first polymerization step. This allows them to restart the polymerization at their discretion, thereby allowing the second polymerization step to be selectively localized around the solubilized nanoparticle.
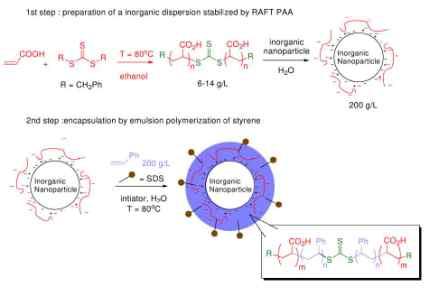
It is intriguing that the authors' process does not rely on a very strong adsorption of the initial PAA coating on the particle surface. A strong interaction does lead to the core-shell structure, but it is apparently not required. Instead, if there is only a comparatively weak interaction between the PAA and the particle, then as the second polymer grows it becomes more and more hydrophobic, and it is therefore trapped more and more strongly on the particle--effectively being pressed onto the surface by its unfriendly aqueous surroundings.
The simplicity, generality, and scalability of this method should make it attractive to manufacturing.
Image from J.-C. Daigle and J. P. Claverie, "A Simple Method for Forming Hybrid Core-Shell Nanoparticles Suspended in Water," Journal of Nanomaterials, Volume 2008 (2008), Article ID 609184. Reprinted under a Creative Commons Attribution License
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported.
