| This study proposes the mechanism of toxicity of starch coated silver nanoparticles in human lung and brain tumor cells is through disruption of the mitochondria, the main source of energy for the cell. Starch was previously shown to make the nanoparticle soluble in water and to protect the nanoparticles from “clumping” in the water or within the cells. An exposure to nanosilver may lead to a decrease in metabolism at the cellular level. Further investigation is needed to determine if silver nanoparticles are safe for biological and medical applications. |
Reviewed by Christie M. Sayes and Vanessa De La Rose, Deptartment of Veterinary Physiology and Pharmacology, Texas A&M University
- AshaRani PV, Low Kah Mun G, Hande MP, Valiyaveetil S. 2009. Cytotoxicity and Genotoxicity of Silver Nanoparticles in Human Cells. ACS Nano 3(2):279-290. DOI: 10.1021/nn800596w
Background
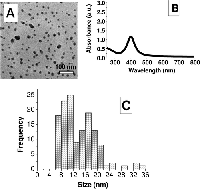
The second potential toxic pathway is due to the silver ions from the surface of the silver nanoparticle. These ions can accumulate in tissues causing metal poisoning or even death. This study was designed to reveal direct disruption of the mitochondria or damage to DNA.
Objective
To assess the mechanism of toxicity of starch coated silver nanoparticles in human lung and brain tumor cells by observing changes in mitochondrial functions and DNA.
Participants/Methods
Toxicity was evaluated using various assays measuring energy production, metabolic activity, DNA damage, and chromatin mutations. Methods involving staining cellular and tissue components were also performed to observe changes in the amount of oxygen used in the cell. Changes in these normal cellular functions would support mitochondrial disruption as a possible mechanism.
Evidence
Data revealed a decrease in energy production as nanoparticle dose and exposure time increased. Because exposure to starch alone did not show significant toxicity, this decrease in energy can be attributed solely to the silver nanoparticle. In addition, the silver nanoparticles produced oxygen radicals. Radicals are atoms, molecules, or ions with unpaired electrons. These unpaired electrons are usually highly reactive and are likely to take part in chemical or biological reactions. In a cellular system, oxygen radicals are known to cause damage to the mitochondria, which was observed in both cell types used in this study.
The authors have shown that there is a silver nanoparticle concentration-dependent increase in DNA damage with a greater prevalence in the cancer cells.
Results
Results of the study indicate starch coated silver nanoparticles may be disrupting the mitochondria, thus decreasing metabolism and increasing damage at the cellular level to both normal and cancerous cells. Specifically, damage to DNA is a predisposing factor in cancer. The authors state that even a small dose of silver nanoparticles has the potential to cause toxicity.
It is essential that the mechanisms of silver nanoparticle exposures be explored before initiating future medical and biological applications.
Image reproduced with permission of AshaRani PV, Low Kah Mun G, Hande MP, Valiyaveetil S. 2009. Cytotoxicity and Genotoxicity of Silver Nanoparticles in Human Cells. ACS Nano 3(2):279-290. DOI: 10.1021/nn800596w . Copyright 2009 American Chemical Society.
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported.
