| Yang et. al. describe a unique nanostructured rechargeable battery, wherein a lithium sulfide (Li2S)-mesoporous carbon cathode and silicon nanowire anode are combined to form a full cell. By incorporating the lithiated counterpart, Li2S, in the cathode, issues associated with elemental lithium anodes can be avoided while retaining the high capacity and performance offered by the silicon nanowire anode. This work provides an excellent example of the impact of nanostructured materials and emerging nanomanufacturing methodologies in extending the performance of specific materials systems for rechargeable battery applications. |
Reviewed by Jeff Morse, PhD., National Nanomanufacturing Network
Research and development of rechargeable battery materials and technologies has experienced renewed emphasis in recent years due to the increased performance requirements for emerging portable electronics, as well as the impending applications for electric vehicles and grid scale energy mediation for renewable power sources. As a result, major developments in this area will provide significant societal impact and provide substantial market opportunities. Leading the rechargeable battery developments for over a decade now are lithium ion (Li+) battery chemistries that have outpaced other competitive materials systems in cost, performance, lifecycle, and environmental impact. While performance improvements in Li+ technologies have kept pace with requirements for many consumer electronics, they remain insufficient for many future applications such as electric vehicles due to limited specific charge capacity of the electrode materials.
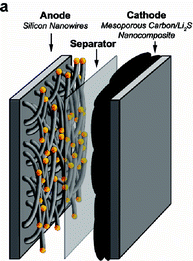
New paradigms presented by emerging nanofabrication methods combined with nanostructured materials compositions enable previously investigated materials systems to be re-evaluated. One such example is the lithium/sulfur system where the redox reaction 2Li + S => Li2S has the potential to overcome the limitations of present cathode materials--sulfur cathodes having a theoretical capacity several times higher (1672 mAh/g) than present materials systems. Yet sulfur based cathodes are limited by several factors, including low electrical conductivity, structural and volumetric changes during charge/discharge cycling, and dissolution of polysulfides in the electrolyte, thereby diminishing capacity over charge cycles. These issues were recently improved through the use of a nanocomposite sulfur/mesoporous carbon material system demonstrating a cathode capacity >1000 mAh/g, along with selective surface coatings, which further stabilized the sulfur during charging providing reasonable cycle lifetime. (see Nanostructured Cathodes for Improved Energy Storage in Rechargeable Batteries, 8/21/09).
In these examples of high capacity anode and cathode materials systems, elemental lithium electrodes are typically used in conjunction with standard liquid electrolytes to benchmark the electrode performance. In practice, the use of elemental lithium remains a significant safety concern due to the formation of lithium dendrites during charge cycling that can penetrate the separator resulting in thermal runaway. Therefore, an integrated full cell exploiting emerging high capacity anode/cathode materials systems must incorporate the lithium source without significantly degrading the capacity advantages enabled by the nanomaterials approach.
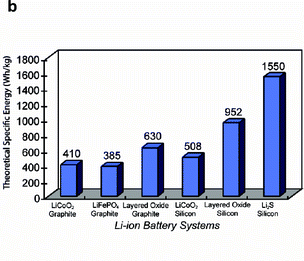
Test results demonstrated a first discharge specific energy of ~630 Wh/Kg based on the mass of the active electrode materials, which was comparable to commercially available LiCoO2/graphite based systems when all materials in the structure were considered. The authors estimate a specific energy for optimized Li2S/Si battery configurations in excess of 600 Wh/Kg as the materials synthesis processes are improved. Furthermore, the integrated full cell structure exhibited ~40% decrease in capacity during charge cycling before stabilizing. Further improvements in immobilizing the Li2S in the mesoporous carbon structure through the application of functionalized surface treatments will minimize the dissolution of polysulfides within the electrolyte, thereby stabilizing the sulfur cathode. While further investigations will lead to an improved understanding of the electrochemical properties of these materials systems, this work provides an excellent example of the impact of nanostructured materials and emerging nanomanufacturing methodologies in extending the performance of specific rechargeable battery designs and applications.
Images reproduced with permission from Yang Y, et.al. 2010. Nano Letters 10(4): 1486–1491. DOI: 10.1021/nl100504q. Copyright 2010 American Chemical Society.
This work is licensed under a Creative Commons Attribution-NonCommercial 3.0 Unported.
