| Organic photovoltaics represent an attractive approach for the production of cost competitive solar cells, as they incorporate solution-based processes that are compatible with large-area, high-throughput manufacturing infrastructure. Enhanced device performance can be achieved incorporating organic/inorganic blends to form hybrid BHJ solar cells with improved electron mobility, dielectric constant, photon absorption, as well as enhanced physical and chemical stability. In this study, a solution-based method has been investigated to fabricate a new type of hybrid BHJ solar cell. By achieving self-alignment of semiconducting NW structures within the active layer, performance improvements were realized through enhanced carrier extraction pathways. |
Reviewed by Jeff Morse, PhD., National Nanomanufacturing Network
Organic photovoltaics represent an attractive approach for the production of cost competitive solar cells, as they incorporate solution-based processes that are compatible with large-area, high-throughput manufacturing infrastructure. One of the drawbacks associated with these photovoltaics, which are also referred to as bulk heterojunction (BHJ) solar cells, is that the polymer blends—mixtures of conjugated polymers and C60 derivatives— form random networks of electron acceptor/donor regions within the active area of the cell, resulting in somewhat inefficient exciton dissociation and charge collection pathways. In order to improve the efficiencies of these processes, an ordered, interpenetrating network of electron acceptor and donor regions having domain sizes on the order of 10-30 nm matching the exciton diffusion length are ideal. Further enhancements in device performance can be achieved incorporating organic/inorganic blends to form hybrid BHJ solar cells with improved electron mobility, dielectric constant, photon absorption, as well as enhanced physical and chemical stability. The inclusion of inorganic nanoparticles such as TiO2 or ZnO in a hybrid BHJ cell is still limited in performance by the charge hopping transport through the discontinuous percolation pathways.
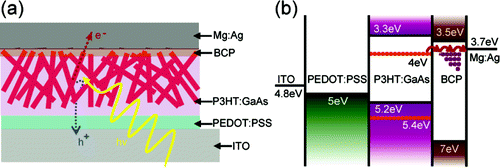
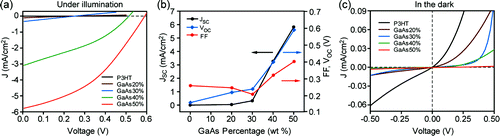
Devices having GaAs NW weight percentages of 0, 20, 30, 40, and 50% in the active layer blend were fabricated and characterized. Under white light exposure, the authors observed a threshold limit of 30 wt% GaAs NW loading where the short circuit current begins to increase. This loading correlates with the formation of GaAs NW percolation networks, thereby enhancing the extraction of charge from the device. A similar trend was observed for cell open circuit voltage and fill factor. Additionally, the dark leakage current decreases with NW loading. The authors attribute these performance enhancements to an increase in the molecular ordering in the P3HT phase with increased NW loading. Characterization of the active layer film by techniques including TEM, AFM, and grazing incidence X-ray spectroscopy indicated some level of vertical alignment of the NWs, with average spacing of ~35 nm for the 50 wt% NW loaded films. To further enhance cell performance, the authors engineered the interface between the P3HT and NW to better facilitate charge separation by preparing GaAs-TiOx core shell NWs by coating the GaAs NWs using a solgel chemistry. The power conversion efficiency for cells incorporating the core-shell NWs was measured to be 2.36% at 50 wt% NW loading, an increase of 20% over the bare GaAs NWs at similar loadings.
In summary, a solution-based method has been investigated to fabricate a new type of hybrid BHJ solar cell. By achieving self-alignment of semiconducting NW structures within the active layer, performance improvements were realized through enhanced carrier extraction pathways. The molecular ordering of the conjugated polymer facilitated by the one-dimensional NWs at loadings exceeding the percolation threshold can potentially provide new approaches that can be extended to other systems having one-dimensional elements. Additional investigations of the hybrid BHJ solar cell devices might include better matching of the semiconducting NW bandgap with that of the conjugated polymers.
Images reproduced with permission from Ren S, et al. Nano Letters Article ASAP 20 December 2010. doi:10.1021/nl1030166.Copyright2010 American Chemical Society.
This work is licensed under a Creative Commons Attribution-NonCommercial 3.0 Unported.
